Healthcare providers are always welcome to consult with our Family Services Team about specific issues so they can care for patients with Tay-Sachs, Canavan, GM1 gangliosidosis, and Sandhoff diseases.
Skip to any section using the links below:
Overview
National Tay-Sachs & Allied Diseases Association (NTSAD) is dedicated to finding a cure for Tay-Sachs, Canavan, GM1 gangliosidosis, and Sandhoff diseases. We support researchers around the world through our Research Initiative, collaborative programs, and fundraising. The Scientific Advisory Council (SAC) supports our efforts by reviewing research proposals, monitoring the field, and sharing important developments and breakthroughs.
On this page, you’ll find information on the research process and how to participate in clinical trials, natural history studies, and patient registries.
If you’re looking for information on a specific disease, please visit Diseases.
About the Research Process
We support researchers who are working hard to develop treatments and ultimately a cure for Tay-Sachs, Canavan, GM1, and Sandhoff diseases.
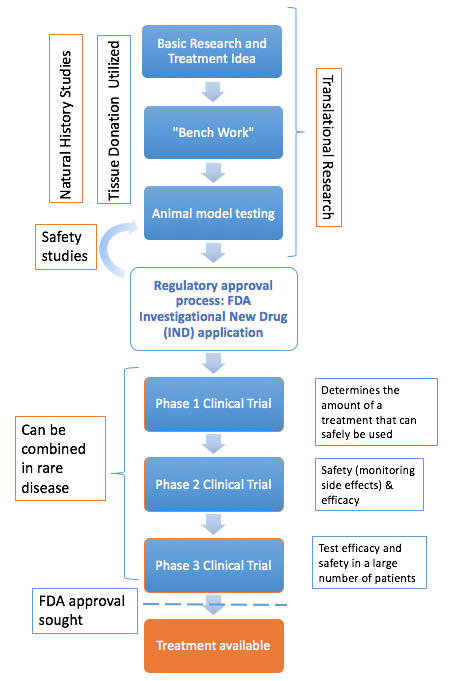
This is a long and complicated process that begins with basic research to understand what causes the disease and how it progresses. At this stage, the basic research is informed by natural history studies and tissue donation. Researchers develop theories about how a treatment could work and test these theories in a lab. The treatments are tested using animal models and, if they work, they undergo a rigorous preclinical approval process to obtain approval by the Food and Drug Administration (FDA).
If a treatment is deemed safe, clinical trials begin. Clinical trials usually have three phases that test safety and dosage, effectiveness, and potential side effects separately. For rare diseases, the FDA will often allow the first two phases to be combined because there is a limited number of potential candidates.
After clinical trials, the treatment seeks FDA approval to be used to treat the specific population it was tested in. Once approved, clinicians can prescribe the treatment to patients in the United States.
How Can I Take Part?
The decision to take part in a clinical trial, natural history study, or tissue donation is intensely personal. We support individuals and families in the process of making those difficult decisions.
Clinical Trials
In clinical trials, participants will receive a treatment to test whether it is safe and effective, and to determine the treatment’s side effects. Researchers use information provided through natural history studies to determine whether the treatment is effective.
Natural History Studies
Natural history studies observe the course of a disease as it progresses over time. There are no treatments or interventions associated with the studies.
The studies present no risk to participants, and they benefit the entire Community. The information collected provides baselineBaseline is a technical term for “the new normal” or how things looks when they are stable. With Tay-Sachs, Canavan, GM1 gangliosidosis, and Sandhoff diseases, the baseline shifts, and often it shifts down over time like steps, where each lower step represents a lower level of functioning. More knowledge about the disease progression, which researchers can use to measure the effectiveness of various therapies during clinical trials.
Tissue Donation
The donation of brain tissue provides researchers with the invaluable opportunity to learn more about a rare disease. In advancing their understanding, researchers are better equipped to develop effective treatments and eventually, a cure.
Need Help?
If you have questions or need more information about research, clinical trials or studies to support your individual or family decisions, please email Diana (diana@ntsad.org), Director of Family Services, or Becky (becky@ntsad.org), Family Services Manager.